Although the meeting was billed as a scientific conference, a number of anti-evolutionists were also in attendance, including several people associated with the Center for the Renewal of Science and Culture (CRSC), the creationist arm of the Discovery Institute, a Seattle-based organization that advocates "intelligent design" as an explanation for biotic diversity. Indeed, one of the principal organizers of the meeting was Paul Chien, a marine toxicologist at the University of San Francisco in Santa Rosa, California, and a senior fellow of the CRSC. The talks were scheduled to provide prominent slots for CRSC fellows and their associates. Even more troubling was the fact that scientists were not informed of the involvement of the CRSC before arriving at Chengjiang and only became aware of its involvement once they inspected the printed abstracts of the presentations. Many scientists are becoming concerned about the activities of the CRSC, and so it seems important to clarify what occurred at this conference.
My experience at the meeting convinces me that all the anti-evolutionists who attended were motivated by political, not scientific, interests. There is nothing inherently wrong with that, of course, although it is markedly unusual in a scientific conference. I even had to admire the nerve of several anti-evolutionist speakers who made presentations in front of opponents as formidable as Eric Davidson, the developmental biologist from the California Institute of Technology and a member of the US National Academy of Sciences. Nevertheless, I could find nothing in any of their presentations that provided scientific evidence suggestive of the action of an intelligent designer, undiscovered natural laws that govern the development of form, or the action of some unspecified principle of "harmony" that drove the early evolution of animals.
What was presented were old Paleyian arguments for design cast up in a variety of molecular guises. These arguments are based on the notion that, because we do not currently understand all aspects of the evolution of life, evolutionary ideas must therefore be fundamentally flawed and, therefore, there must be an intelligent designer. Rather than presenting a coherent argument for the action of an intelligent designer, these advocates were more interested in exploring what they present as weaknesses in evolutionary thinking. Their position ignores the colossal amount of concordant evidence supporting evolution and refuses to acknowledge the legitimate complexity of modern evolutionary thinking.
Unlike the contingent of scientists, the anti-evolutionists at the meeting all appeared to have known of the CRSC's involvement with the meeting before their arrival. These anti-evolutionists represented a broad range of anti-evolutionary viewpoints. At one extreme their camp included Michael Denton, a senior fellow of the CRSC, whose professed "pagan" personal philosophy, as he explained it to me, seemed about as far from biblical literalism as one could imagine. At the other extreme there was a young, fervently Christian student who spoke candidly of his belief in young-earth creationism. Denton is a qualified geneticist, and I could trace no scientific or philosophical link between his platonic notions of natural laws that govern the form of animal archetypes and the strict biblical-literalist, young-earth stance espoused by the student. The fact that both these individuals claimed that the Discovery Institute had financially supported their attendance at the meeting suggests that the Institute's involvement in the meeting was not motivated primarily by the desire to present a coherent scientific argument for "intelligent design". Rather, it suggests that the Discovery Institute was more interested in supporting any views that appear to challenge evolutionary explanations, regardless of whether these views are mutually exclusive.
Although I cannot read the minds of the individuals associated with the CRSC, I can advance a reasoned interpretation of what might be motivating their interest in this issue. The US Supreme Court ruled in Edwards v Aguillard (1987) that creationism cannot be taught in science classes in public schools because it is not a scientific concept and because there is no secular purpose in teaching it. Advocates of intelligent design creationism (IDC) may attempt to circumvent this ruling by arguing that "intelligent design" is a scientific alternative to evolution. Tactics for doing so might include having IDC representatives speak at scientific meetings alongside recognized scientists and having their ideas published alongside scientific papers in conference proceedings. The association of "intelligent design" with legitimate scientific conferences and publications could be presented to US lawmakers as evidence that advocates of IDC are pursuing reputable science rather than sectarian politics, and that the scientific community accepts "intelligent design" as a viable and productive topic of study. This in turn would support the recent debate about "alternatives to evolution" to public school science classes. The remainder of this article seeks to identify specific positions that those associated with the Discovery Institute appear to take with regard to scientific evidence on this matter, and then respond to these positions from my own viewpoint as a scientist.
Why Anti-evolutionists Focus on the Origin of Animals and The "Cambrian Explosion"
It seems likely that the origin of animals will remain a favorite subject for anti-evolutionists over the coming years. Before reviewing the current scientific evidence concerning the origin of animals, I wish to outline the strategic position of IDC anti-evolutionists — especially those associated with the CRSC — with regard to the origin of animals as far as I understood it from the meeting.IDC advocates claim that: 1) The major groups of animals had separate, independent origins (by "major groups of animals" anti-evolutionists mean the marine creatures without backbones that commonly correspond to the "invertebrate phyla" such as Mollusca, Brachiopoda, Arthropoda, and so on ... in addition to the first Chordates). This position, of course, denies common ancestry among living taxa. 2) These major animal groups originated over a "very short" interval of geological time associated with the "Cambrian explosion" (some IDCs suggest a period of 2-3 million years). This is much shorter, of course, than expected by most evolutionary models.
These two points lead to the conclusion that the rate and magnitude of innovation were far too high to be accounted for by natural selection and can only be explained as the actions of a designer.
The IDC position is challenged by the scientific facts. The claim that the major groups of animals appear suddenly in the fossil record is easily demonstrated as incorrect by the extensive fossil record of early animal evolution that stretches back several tens of millions of years earlier than the Chengjiang fossil beds. So anti-evolutionists are in deep denial of the fossil record when they cite the impossibly short interval during which new taxa emerged.
The claim that the major animal groups originated separately and independently is equally weak. The origin of animals took place relatively late in the earth's 4.6-billion-year history; however, recent fossil discoveries contain evidence of a long and rich ancestry for animal phyla. Of course, when most people think of the "major groups" of animals, they envision the vertebrate classes that represent what first think of as "animals" today — mammals, reptiles, amphibians, fishes, and birds. Vertebrates represent only some of members of one phylum — the Chordata. There are numerous other phyla, represented by such disparate creatures as starfish (Phylum Echinodermata), spiders and crabs (Phylum Arthropoda), clams and snails (Phylum Mollusca), in addition to many others.
In a broad sense each different phylum has a distinctly different fundamental structure and development — and each represents a different "body plan", or set of "body plans". Scientists understand body plans to refer to the major features of adult bodies in metazoans and/or of the developmental trajectory that gives rise to the adult body. Different body plans are distinguished on the basis of variation in features such as aspects of skeletal construction, symmetry, internal body cavities, segmentation patterns, and appendage structure.
Almost all metazoan phyla can be instantly distinguished from one another on the basis of variation in these fundamental features. The animal taxa that emerged out of the Precambrian are clearly related to living taxa, but even the early chordates — the founding members of the phylum that would later give rise to the vertebrates — bore little superficial resemblance to the vertebrates that would finally appear 100 million years later.
The IDC conclusion that the Cambrian Explosion can be explained only by reference to an intelligent designer is unsupported by the scientific evidence, as discussed below. Moreover, the common anti-evolutionist strategy of quoting Darwin as if science has stood still since he wrote On the Origin of Species backfires. One of the most remarkable aspects of the Origin is the way in which Darwin identified potential criticisms of his theory and addressed them with refreshing honesty. He was very frank about what he thought was the absence of fossils in rocks older than those bearing the oldest skeletonized fossils known in Europe (now known to be about 525 million years old), recognizing this absence as a "valid argument against the views here entertained" (Darwin 1859 [1964: 308]). After admitting that he had no solid explanation of the absence of these fossils, Darwin advanced some hypotheses about the incompleteness of the geological record. It is instructive to compare what we now know about the early history of life on the planet with what was known in Darwin's time and to ask how his views have stood the test of time.
The scientific issues relating to the origin of animals and the Cambrian Explosion
What defines an animal, and how are different animal groups related?
The time around the Precambrian-Cambrian transition is important because it provides us with the first fossilized record of metazoans — multicellular animals with features such as differentiated organs and tissues — about 544 million years ago. I emphasize that "appearance" is not the same thing as "origin". There are myriad reasons related to the preservation and recovery of fossils that can explain why the first recorded appearance of a particular group can occur substantially after its evolutionary origin. The origin of animals is a complex issue for which several independent lines of evidence need to be investigated.First, we need to decide what it actually is to be an animal. To do so, we must identify unique characters that are shared by all animals and distinguish them from other types of organisms. These are the characters that diagnose animals as a natural group and are considered ancestral for all animals. Once we have done that, we can proceed to identify other novel characters that distinguish specific subgroups (or "clades") of animals from the ancestral state and from each other. This process of distinguishing groups within groups produces a hierarchical nested set of related animals.
Animals are multicellular organisms that have cells specialized to perform particular functions; these cells are held together by an organic glue called extracellular matrix (ECM). On the basis of these features, biologists have long inferred that all animals constitute a natural group and evolved from a single common ancestor. But multicellularity with specialized cells is a general similarity — one that applies to some organisms, such as trees, that we would all agree are not animals — so multicellularity with specialized cells is not enough to prove common ancestry of all animal taxa. These features may have evolved independently in different lineages of single-celled organisms, and so it is the highly specific shared features, such as the nature of ECM, that assume a special significance for defining what it means to be an animal. This is because very specific similarities are unlikely to have arisen convergently and so point towards a single common ancestor for all animals. Recent discoveries of numerous very specific properties shared by all animals provide extremely strong evidence for their common ancestry.
We now know that all animals share not only general similarities but also many highly specific genes, for example, the transcription factors of the ets gene family, paired-box genes, and a primordial Hox gene (Peterson and Davidson 2000). These genes are fundamental in organizing the layout of animal bodies, and have such similar molecular structures that we can confidently conclude that they result from common ancestry, rather than from later evolutionary convergence. Thus they provide extremely strong evidence that all animal groups arose from only one lineage of single-celled ancestors.
The simplest animals, sponges, have all the characters mentioned above (along with a few unique characters of their own), but they lack the next set of features that diagnose an evolutionarily clade of animals derived somewhat later. In accordance with the nested hierarchy of characters we expect, all animals, except the sponges, to exhibit embryonic gastrulation (a special infolding of the wall of the initial ball of cells formed after fertilization), and the duplication of the primordial Hox gene. These features form the basis for diagnosing a more derived group of animals that includes corals and all other metazoans (ourselves included), but excludes the more basal clade that contains the sponges. At each step of the evolution of animals, we can demonstrate a similar diagnostic branching.
What we have just done is to use the distribution of novel features to map the evolutionary changes that both diagnose what makes an animal in the first place and tell us how animals within the group are related. The resultant hierarchical nested sets of related animals are exactly what we would expect according to an evolutionary model. By any reasonable evaluation, these must be considered strong evidence of the evolutionary relatedness of all animals. Of course, the spate of new information on molecular structures and developmental genetics raises many new questions, but the big picture is that these new data can only be viewed as furnishing wonderful vindication of Darwin's central notions. Time and time again, we find that animal subgroups thought to be related on the basis of morphological evidence also share exclusive similarities in gene sequences and in patterns of developmental control, as predicted by evolutionary theory. At the Chengjiang meeting, the CRSC's Jonathan Wells suggested that developmental genetic evidence favors separate origins from different single-celled lineages for the major animal groups. But his suggestion contradicts a wealth of scientific evidence and therefore must, in my view, arise from non-scientific convictions.
Our understanding of the major relationships among animal groups is now stabilizing. This is not to say that we currently know all there is to know about animals' relationships or molecular biology — far from it, which is why evolutionary biology is an exciting area of research. Anyone with knowledge of developmental biology and sufficient time can find aspects of specific systems or pathways the operation and evolution of which are not now fully understood. But what we do understand strengthens the case for evolution because new insights and methods of analysis, unthinkable in Darwin's day, yet again fulfill the predictions of the evolutionary model.
What are the implications of the pattern of relatedness discussed above for evolution and for intelligent design creationism? First and most important, it clearly falsifies the IDC claim that animal groups appeared separately and independently. Instead, we see a hierarchy of the distribution of shared features — some general to the group as a whole, others specific to particular subgroups. Such a distribution of features is predicted by evolutionary theory, which was proposed long before most of these features had been recognized. It is not concordant with the idea of multiple independent origins of animals, because that model would have no compelling basis on which to predict a hierarchical arrangement of such shared features.
An apologist for "intelligent design" could argue that a designer worked sequentially in a series of small steps, which could explain why the features defining clades are arranged hierarchically. Curiously, however, members of the CRSC apparently do not apply this explanation to the Cambrian biota. Rather, they persist in asserting the independent origins of different animal groups, despite overwhelming scientific evidence against that viewpoint.
The extensive Precambrian fossil record
The second pillar of the IDC position — that major groups of animals appeared too quickly for natural processes to account for them without invoking the intervention of an intelligent designer — is equally unsupported by the scientific evidence. However, to expose the weakness requires some background information about the Precambrian-Cambrian transition.The earliest fossils currently known occur in rocks from western Australia that date from around 3465 million years ago (Shopf 1993), and a reasonably good fossil record is known from that time onwards. The earliest chemical evidence of life itself is even older, about 3900 million years ago (Mojzsis and Harrison 2000). The earliest fossils are prokaryotic cyanobacteria, and as we move up through the geological column toward the base of the Cambrian these forms are joined by more complex fossils, such as those of eukaryotic cells, by about 1800 million years ago (Knoll 1992). (It is important to reiterate that the first occurrence of a fossil marks the minimum age for the appearance of the group to which it belongs, but the origin of the group often is far earlier, as Darwin suggested. For example, there is good chemical evidence that eukaryotes existed from about 2700 million years ago [Brocks and others 1999], but the earliest fossils yet found that are widely accepted as eukaryotes are some 900 million years younger.) The increase in complexity and diversity of fossils through the Precambrian up toward the boundary with the Cambrian is concordant with an evolutionary explanation, and the sequence of appearance makes sense in evolutionary terms. Darwin would be justified in feeling vindicated by these discoveries of definitive Precambrian fossils, which were unknown at the time he was writing. Anyone who suggests that Darwin's 19th-century difficulty with an apparently abrupt start to the fossil record still pertains today simply has not considered the evidence.
When and how quickly did animals first appear, and did all "major groups" appear at the same time?
We do not yet know exactly when the first animals originated because we do not yet know exactly when the definitive characters of animals — extracellular matrix, the primordial Hox gene, the ets gene family, and so on — originated. There is a wide range of estimated dates for the differentiation of the major groups of animals from one another. Some studies suggest that this occurred as much as 1100 million years ago; others suggest a date closer to 600 million years ago (see Valentine and others 1999).Although the methods used in these estimates are not currently as precise as they may yet become, it is hardly a surprise that the dates for the divergence of major groups are spread over a wide timespan. This is because major groups of animals are related in a hierarchical fashion, as we saw above, and would thus be expected to diverge from an ancestral lineage at different times. Therefore, we would expect the split between vertebrates and echinoderms, groups that share a wide range of derived features, to have occurred more recently than, say, the split between sponges and the common ancestors of echinoderms and vertebrates. Why? Because sponges are among the most basic animals with the fewest derived features, and so we would expect them to have split off earlier. And this is exactly what we do find — sponges and other animals are estimated to have separated about 950 million years ago, whereas echinoderms and vertebrates split from each other somewhere between 700 to 550 million years ago (Smith 1999).
Three lines of evidence provide important constraints on estimates of when the key events in animal evolution happened. Although the three lines of evidence are independent of one another, the results of each approach are concordant with the inescapable conclusions that the origin of animal groups was a protracted affair that required at least 100 million years and possibly far longer, and that the origin of animal groups took place in the Precambrian, long before the "Cambrian Explosion". Thus the IDC position that the origin of animals occurred very quickly as part of the Cambrian Explosion is falsified by these lines of evidence.
The three lines of evidence are:
1. Molecular clocks. If we can estimate the rate at which particular organic molecules change among living groups whose divergence times are well known, then we can compare the amount of difference in the same molecules among a wide variety of animals to calculate approximately when these forms diverged. The estimated rate of change in these molecules is the basis for molecular clocks, and the relationships suggested by multiple molecular clocks demonstrate concordant patterns. We now know that the major novel characters that distinguish the major groups of animals appeared at most 1500 million years ago, but at the latest no more than about 560 million years ago — 15 million years before the start of the Cambrian Period (Lynch 1999; Smith 1999), and about 40 million years before the age of the fossils of Chengjiang.
2. Evidence from body fossils. There is a substantial Precambrian fossil record of animal bodies and body parts. For example the calcareous tube Cloudina represents the outer skeleton of an animal, and has long been known from Precambrian rocks at least 550 million years old. There is undisputed evidence of fossil sponges dated about 545 million years ago (Brasier and others 1997), right about at the Cambrian boundary (544 million years ago), but there are also fossils of sponge embryos dated at around 580 million years ago (Chen and others 2000). Some scientists have also argued that some members of the Ediacaran fauna — an enigmatic suite of late Precambrian body fossils about 555 million years old — represent a variety of animal groups with representatives living today. There is widespread agreement that at least some of these forms represent sponges or cnidarians (jellyfishes, corals, sea anemones, and hydras), but some scientists argue that arthropods and mollusks are also present in the Ediacaran assemblages. Moreover, Chen and others (2000) recently claimed to have recovered embryos similar to those of derived groups such as arthropods and echinoderms in deposits about 580 million years old. These researchers argue that early animal evolution took place at small, almost microscopic sizes, unlikely to leave much of a fossil record. If these interpretations of the Precambrian fossil record are correct, they strengthen the case for argument that the differentiation of the "major groups" occurred much earlier than their dramatic appearance in the Cambrian Period.
3. Evidence from trace fossils. Trace fossils are evidence of the activity of animals, such as the burrows, tracks and trails that animals left on the sediment surface or beneath it in the seafloor. There is no serious argument that large trace fossils were formed by anything other than animals, although there is some debate as to which are the earliest trace fossils, because many simple trace fossils, such as might be formed by the earliest animals, are easily confused with other structures produced by inorganic processes. One thing is very clear: there are many trace fossils in Precambrian rocks at least 555 million years ago, and possibly far earlier (Budd and Jensen 2000). It is also clear that the order of appearance of trace fossils proceeds sequentially from simple to more complex forms (Budd and Jensen 2000). Indeed, one feature that identifies the Cambrian period in the geological is the appearance of the burrow network Treptichnus pedum. The distinctive form of this trace leaves no doubt that it was formed by an animal with a central gut and a reasonably sophisticated neural system. T pedum first appears some 10 million years before skeletonized fossils become common and about 20 million years before the Chengjiang fauna lived (about 525 million years ago).
What does all this mean? The lineages that include the major groups of animals (each major group being characterized by a particular "body plan") certainly diverged during the late Precambrian and not during the Cambrian itself. Because major animal groups share so many developmental features, these features must have originated before these lineages split — that is, at least 580 million years ago, some 45 million years before the beginning of the Cambrian Period. Fossil evidence, both from trace and body fossils, is consistent with this interpretation, and the trace-fossil record suggests the stepwise acquisition of increasingly complex behaviors from about 555 million years ago onward. Hence, any suggestion that the appearance of the first representatives of "modern" animal groups in the Cambrian correlates specifically with the time of origin of these groups (an argument favored by proponents of IDC) is clearly refuted by the evidence, which shows that the major groups of animals originated during the Precambrian.
It is also important to appreciate that about 20 million years pass from the beginning of the Cambrian to the time of the Chengjiang fauna — the Burgess Shale fauna (discussed in Gould's Wonderful Life [1989]) is even more recent. Because Treptichnus pedum, the marker for the start of the Cambrian, was made by an animal with a gut and complex behavior, it is certain that large animals with 3 layers of cells (the triploblasts) were living long before the debut of the stars of Wonderful Life. Any suggestion that animals evolved within "a mere 2 or 3 million years" (Heeren 2000) of the Chengjiang fauna is an irresponsible and bizarre misrepresentation that flatly contradicts scientific facts.
What, then, does the Cambrian Explosion represent?
The Chengjiang fauna, like that of the Burgess Shale and several other Cambrian sites, is truly remarkable for the quality and the range of biological diversity that it preserves. Detailed work on these faunas has revealed a remarkable fact — that animals related to the major living groups of animals were present from at least the later portion of Early Cambrian Epoch. In Wonderful Life Gould (1989) made much of this important fact by suggesting that because forms comparable to major groups of living animals were already present in the Cambrian, later evolutionary history has mostly involved variation on established themes, rather than the origin of really major new animal body plans. He also stressed that several fundamentally distinct animal body plans present in the Cambrian have since vanished. These body plans are found both in lineages that belong to existing phyla (for example, some extinct groups of Arthropoda) and in lineages that seem allied to other metazoans but are obviously not members of living groups (for example, the Archaeocyatha). Thus, according to Gould, most of the fundamental innovations in body plan were in place by Middle Cambrian time, and the Cambrian fauna was more diverse than its modern counterpart.New discoveries and interpretations in paleontology and developmental genetics have changed the scientific landscape significantly since Gould wrote Wonderful Life. Wills and others (1994) have suggested that Gould may have overestimated the diversity of Cambrian animals, although scientists disagree on how best to measure this diversity. Nevertheless, Gould's central point — that at least some groups of Cambrian animals exhibit a morphological diversity that is at least comparable to that seen in living fauna — remains valid and should not be underestimated. Compared with the fauna of the later Precambrian, the Cambrian fauna is strikingly diverse; the recent discoveries of early vertebrate-like fossils in the Chengjiang beds simply emphasize the point that much innovation was in place by relatively early in the Cambrian Period.
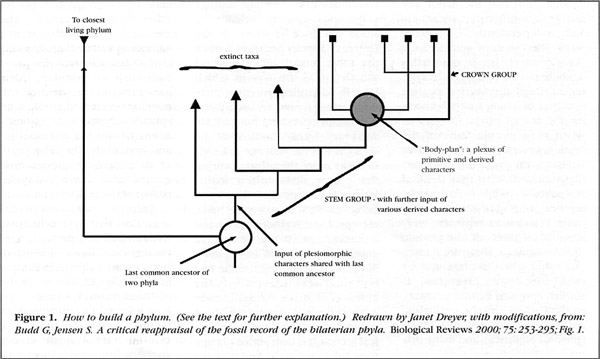
The aspect of Gould's views that has been most strongly challenged is the idea that several fundamentally distinct animal body plans have vanished since the Middle Cambrian. The definition of what constitutes a "fundamentally distinct animal body plan" is difficult because it requires an evaluation of the evolutionary "weight" or significance of particular features; we are not yet sure how to assess this weight objectively. But what has become clear recently is that the Burgess Shale and Chengjiang faunas contain not only members of "crown" groups (those with living representatives), but also animals in "stem" groups, which are more distant relatives of these surviving groups (Budd and Jensen 2000).
Because species belonging to stem groups are typically extinct, their place in the phylogenetic tree can be difficult to interpret. It turns out that many of the forms Gould interpreted as representing additional fundamentally distinct body plans may merely be evolutionary adventures or "experiments" within the lineages of the major groups of animals we know today. So, the unusual fauna of Chengjiang and in the Burgess Shale likely represent way stations along the road to the establishment of the modern groups rather than cul-de-sacs of evolutionary innovation. Some forms we can easily recognize as linked to living groups; others are more enigmatic (and stem groups, of course, show evidence of transitional states). But such difficulty in interpreting early fossils, of course, is what Darwin predicted in the Origin of Species, because he knew that the selective action of extinction throughout geological time could only tend to emphasize differences, not similarities, among these major lineages of animals.
The bottom line is that the establishment of modern animal groups was a protracted affair that began no later than about 600 million years ago, extended across the Precambrian-Cambrian boundary, was still in progress during the Early Cambrian Epoch, and continued after the close of the Cambrian Period. Accordingly, science currently tells us that there was, at a minimum, about 100 million years from the time when the first sponge-like animals originated until the origin of representatives of all the major living lineages or body plans.
Because animals did not evolve in a geological instant, there is no need to invoke some novel evolutionary — or supernatural — process to explain their appearance. This is not to say that the appearance of every novel feature is of the same importance for later evolution or that the rate of appearance of novel features was constant throughout the entire interval. Scientists do not demand such restrictions, even though anti-evolutionists frequently present them as basic premises of evolutionary explanations.
It is clear that there was a fundamental transition that took place over an extended interval across the Precambrian-Cambrian boundary. That transition reflects a dramatic shift in the structure of the ecosystems of early animals, which must have been at least partly fueled by the appearance of new biological innovations. Although a rich mixture of modern groups and their early relatives may have persisted throughout the Cambrian, there is a clear contrast between these generally familiar forms and the more enigmatic fossils from the Precambrian, such as those from Ediacara in Australia. It is clear that the transition into the Cambrian marks a pivotal time in life history. But although much tinkering went on in the Cambrian and thereafter, the most fundamental steps in the origins of animal groups took place during the Precambrian.
To return to Darwin, how does our current knowledge affect his "difficulty" with the "Sudden appearance of groups .... in the lowest known fossiliferous strata"? The answer is clear: Darwin's difficulty has evaporated. We have now identified thousands of fossils that appear earlier in the fossil record than the point at which Darwin thought it suddenly began. Darwin suspected that the impression of sudden appearance was false, and speculated that the false impression was due to the poor preservation of the older rocks in Europe and inadequate attention given to the fossil record. He has now been vindicated. The sequence of appearances that we now know is consistent with evolution, and the additional support for the common descent of animal phyla from new lines of inquiry that even Darwin could not have imagined only reinforces the brilliant predictive power of his insight. Today scientists might quibble about whether he assumed constant of rates of evolution with regard to the origin of animals. But despite this disagreement over rates of change, these fossils show that Darwin was right to suggest that life had been around for far longer before the beginning of the Cambrian than it has been since. As a result, it is appropriate to think of the Cambrian as a period of great phylogenetic diversification — what scientists call an evolutionary "radiation."
The Precambrian/Cambrian radiation and creationism: the original spin
Animals are incredibly complex and wonderful, and understanding their early evolution requires a full consideration of many different lines of evidence. Much is known, new data are appearing at an unprecedented rate, and yet many questions still excite our scientific curiosity. Scientists such as Stephen Jay Gould and Simon Conway Morris are valiantly striving to make these exotic animals and abstruse issues accessible to the public. It is a privilege to be able to witness all this excitement.It is perhaps inevitable that those motivated by a nonscientific agenda will seek to extract snippets and sound bites from the scientific arguments, package them out of context, and feed them to the general public. This is what Fred Heeren did. Heeren is an anti-evolutionist writer who attended the Chengjiang meeting and then peddled his distorted version of the Cambrian radiation to the popular media, with obvious success (see Heeren 2000, an article in the Boston Globe).
Even if creationist misrepresentation of science is inevitable, it is nevertheless regrettable. Deep time was discovered 200 years ago and is now old news. Almost immediately, scientists recognized the sequential appearance of organisms in the geologic record, demonstrating the development of life's diversity through time, and this has never been seriously questioned in scientific circles. The world will be a better place when its human residents, in the brief flashes of time that each of us is privileged to experience, celebrate what science tells us about our place in nature.
