Insects have often been pests of the creationists' attempts to discredit evolution. The bombardier beetle is a familiar example to the readers of Creation/Evolution (Schadewald, 1986). Peppered moths and insect resistance to pesticides have also drawn creationists' ire. However, these entomological examples of adaptation and natural selection are not the most damaging insect pests for creationists, since fossil insects and the transitions they document provide some of the best examples of evolution.
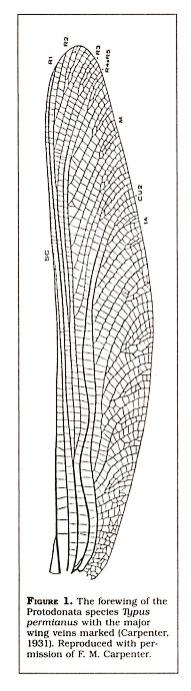
Fossil insects have received little attention in the creation-evolution controversy. Morris discusses the fossil record of insects and claims that there were "giant dragonflies, giant roaches, giant ants and so on. But their form is no different in essence from that of modern insects" (1974). But when the fossil remains of insects are examined, differences from the insects of today are apparent. The "giant dragonflies" were similar dragonflies in that they had large eyes, slender bodies, and could not fold their wings, but they still were very primitive compared to modern dragonflies. These giants belonged to the extinct order Protodonata and had wing spans of between twelve and seventy-five centimeters. Their wing venation (Figure 1) was very primitive when compared to modern dragonflies (Riek, 1970). As another example, fossil roaches included forms that possessed large ovipositors which extant roaches do not possess (Riek, 1970). Finally, there were no giant ants. The oldest fossil ant is from the Cretaceous period and had a head that was just one millimeter in length (Wilson, 1971).
Insects have been preserved as fossils in concretions, amber, shales, and the LaBrea tar pits. The oldest fossil insect dates back to the Upper Carboniferous period. However, six-legged arthropods, such as the springtails (Collembola), have been found in Devonian rocks (Wooton, 1981).
In order to appreciate the significance of the important fossil insects, an understanding of the features used to define the insect orders, suborders, and families is necessary. Unfortunately, an adequate understanding of the various insect groups would require several hundred pages and that is well beyond the purpose of this article. Simply stated, however, the class Insecta contains twenty-seven living and twelve extinct orders (Gillott, 1980; Riek, 1970). The features which define an order include type of mouthparts, form of development, whether or not the insect could fold its wings, and wing venation. Wing venation is very important in classification of the insect orders and families. This is fortunate for our understanding of insect evolution because wings are often the only parts of insects preserved as fossils. Although early entomologists used wing venation to create several extinct orders and families that were not valid, recent work is much more critical. Not only both pairs of wings but also the head and details of the type of mouthparts are required before new ordinal and family designations will be accepted (Carpenter, 1977).
The recent attempt to question the validity of the Archaeopteryx fossil has brought the importance of transition fossils to the forefront of the creation-evolution controversy (Charig et al., 1986). Although Archaeopteryx is possibly the most famous transition fossil, it is by no means the only known transition. There are many fossil insects which fit the criteria of being transitional between higher taxa; that is, they possess features that define two distinct taxa and are found in the appropriate geological period.
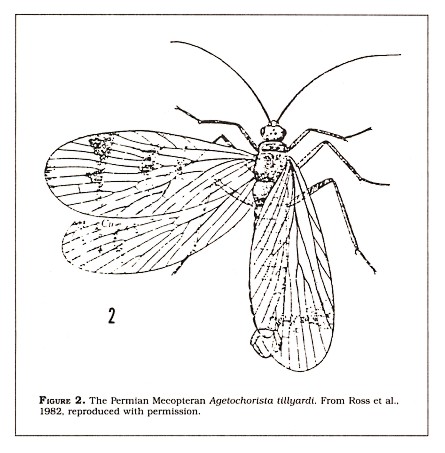
The transition between the Mecoptera (scorpionflies) and the Diptera (true flies) is one of the most impressive insect examples. The Mecoptera today is a small order of nine families with only about four hundred described species. They were, however, much more diverse in the Permian period and in the Mesozoic era with twelve now-extinct families (Ross et al., 1982). Scorpionflies (Figure 2) are small to medium-sized insects with chewing mouthparts often at the end of an elongated head. They also have a characteristic wing venation. The Diptera, on the other hand, are defined as insects possessing one pair of wings, the hind wings having been reduced to small balancing structures called halteres. The wing venation of the Diptera includes a characteristic "kink" near the base of one of the veins. This difference allows for the identification of Mecoptera and Diptera wings. Comparative studies have long suggested that the Diptera evolved from the Mecoptera. The most primitive flies, Tipulidae (crane flies), have features which are suggestive of scorpionflies (Ross et al., 1982; Riek, 1970; Gillott, 1980).
In 1953, E. F. Riek described a Permian fossil wing that had the characteristic kink in the wing vein that is diagnostic of the Diptera. But the wing also had venation that was similar to Permian scorpionflies, so he placed the wing into a Mecopteran suborder he named Protodiptera. The similarity led him to predict that a four-winged fossil fly might be found, and in 1976 he described a more complete fossil of a four-winged insect which possessed the characteristic venation of the Diptera with some of the Mecopteran similarities. Moreover, the hind wings were smaller than the forewings, suggesting that the move toward diptery was on its way. Also, the third segment of the thorax was reduced, another intermediate characteristic between the Mecoptera and the Diptera. The fossil insect, called Choristotanyderus, was used by Riek to define a new suborder of Diptera to include those fossil forms with four wings (Riek, 1976).
The Mecoptera were also apparently ancestral to the order Trichoptera, or caddisflies, mothlike insects which have aquatic larvae and pupae. Like the Mecoptera and Diptera, the Trichoptera have a characteristic wing venation which includes a complete set of longitudinal veins with few cross veins. The Permian fossil Belmontia, which is classified within the Mecoptera, has wing venation which is intermediate between the primitive Mecoptera and the early Trichoptera (Gillott, 1980; Ross et al., 1982). Jurassic fossils show modern trichopteran venation.
These examples are just two of the transitions between orders seen in fossil insects. There are several other fossil insects that yield important information on evolution within the orders. For example, beetles, order Coleoptera, have hardened forewings called elytra. The oldest fossil beetles possess elytra which still retain remnants of wing veins typical of insects without elytra. These first beetles are similar to a group that survives today, the Cupedidae (Riek, 1970). This is an interesting point because these beetles have long been regarded as primitive. The family today consists of only twenty-five species worldwide and is rarely collected, yet it is the first definite beetle to be found in the fossil record and is even found in the early Cretaceous Wealden beds of southern England, suggesting that they were much more common in the past (Jarzembowski, 1984).
Another example is provided by dragonflies, order Odonata. Dragonflies today have a series of wing veins that create a triangular-shaped cell appropriately called the triangle. Harvard's Frank M. Carpenter, using Permian fossils, showed how the wing evolved from those of primitive dragonflies, which did not possess the triangle, to the typical contemporary dragonfly wing with the triangle (1931). This fossil series has important implications for the evolution of dragonfly suborders.
Another case of evolution within an order of insects involves the mayflies, order Ephemeroptera. These delicate insects have membranous wings with many veins. The front wings are large and somewhat triangular-shaped, whereas the hind wings, if present, are small. The mouthparts of living mayflies are vestigial. The mayflies of the past were very different compared to those of today; Paleozoic mayflies possessed forewings and hind wings which were large and equal in size. Moreover, some Paleozoic mayflies had well-developed mandibles (Wooton, 1981).
An excellent example of a family-level transition involves the origin of the family Formicidae, or the ants. The oldest "ant" (Figure 3) is from the Cretaceous amber of New Jersey. It is considered an ant because it has a modified first segment of the abdomen, called the petiole, which is characteristic of ants. E. 0. Wilson and his colleagues who described this fossil were surprised by its wasplike mandibles. In fact, this ant, as they described it in the original paper, "forms a near-perfect link between non-social tiphiid wasps and the most primitive myrmecioid ants" (Wilson et al., 1967).
The Cretaceous amber, as seen with the ant-wasp example, is especially important in providing details of family-level transitions. Another example is the extinct family Jacopidae, which is defined from a Cretaceous amber nymph that is transitional between the families Cercopidae, the spittlebugs, and the Cicadelli, the leafhoppers (Ross et al., 1982). As more work is completed on the Cretaceous amber, it is likely that even more family transitions will be discovered.
As we examine younger amber deposits, such as the Baltic amber from Europe, we find different kinds of transitions. The insects of the Late Eocene show evolutionary transitions below the family level of classification. Many of the genera found in the Baltic amber are still living today, but only one species found in the Baltic amber still survives (Stanley, 1979). These extinct species from the Eocene are providing important details on the evolution of tribes and genera. For example, the ant Agroecomyrmex duisburgi is the "apparent connecting link" between two tribes of ants (Wilson et al., 1967). Moreover, the bumblebees, which belong to the modem genus Bombus, are not found in the Baltic amber or other Tertiary deposits, while the genus Protobombus, thought to be ancestral to the bumblebees, is found in the Baltic amber (Wilson et al., 1967).
It is obvious that there is a trend in the type of transitions that are seen in the fossil record of insects. Clearly, the orders were evolving in the Paleozoic. The evolution of families can be seen in the Cretaceous with the subsequent evolution of tribes, genera, and species occurring during the Tertiary. The finding of transitions within different taxa of insects at different geological periods is not surprising from an evolutionary perspective. Indeed, the value of these insect fossils is best appreciated in the light of a quotation by Duane Gish: "The discovery of only five or six of the transitional forms scattered through time would be sufficient to document evolution" (1978).
Acknowledgements
I thank Dr. Frank M. Carpenter for permission to reproduce illustrations from his extensive work on paleoentomology and Dr. Annette Muckerheide for her critical reading.